Repackaged Avastin® Service
StaClear® syringes now available for the Leiters Health FDA-Compliant Avastin Repackaging Service
Leiters Health was the first 503B outsourcer to introduce and provide repackaged Avastin (bevacizumab) in accordance with the FDA’s 2018 Final Guidance for the Repackaging of Biologics1. In addition to meeting the FDA Guidance we also utilize an advanced filtration process which results in reduced particulates.
As part of our ongoing commitment to quality we are pleased to announce the addition of the StaClear syringe to our Avastin repackaging service. StaClear is the first syringe to meet the FDA’s stringent requirements for an intravitreal indication for use (510(k) Number K200242).
StaClear luer slip syringes are now available for the following products:
- F1042, “Econo-Fill” (0.08 mL fill) – provided with a TSK 30G x ½ inch low dead space needle (unattached).
- F1045, “Convenience-Fill” (0.11 mL fill) provides for physician needle choice.
StaClear Syringe Features and Benefits2
- Cleared by FDA for intravitreal injections (510K 200242).
- Meets the FDAs chemical and biocompatibility standards for ophthalmic use.
- Polydimethylsiloxane slip agent is crosslinked and fixed onto the syringe barrel surface, creating an ultra-low particulate.
- Easy-to-read graduation marks.

The Leiters’ Difference Makes a Difference for your Patients3.
- We utilize an advanced filtration process prior to repackaging the Avastin per FDA Guidelines which results in reduced particulates
- A Certificate of Analysis (CoA) detailing the testing is
provided with every shipment. - USP <789> compliant for visible and subvisible particulates.
- 150-day beyond use date (BUD)3
- Compact space-saving tray to maximize refrigeration space
at your facility.
The Leiters Health Convenience Fill Provides Physicians With An Educated Needle Choice
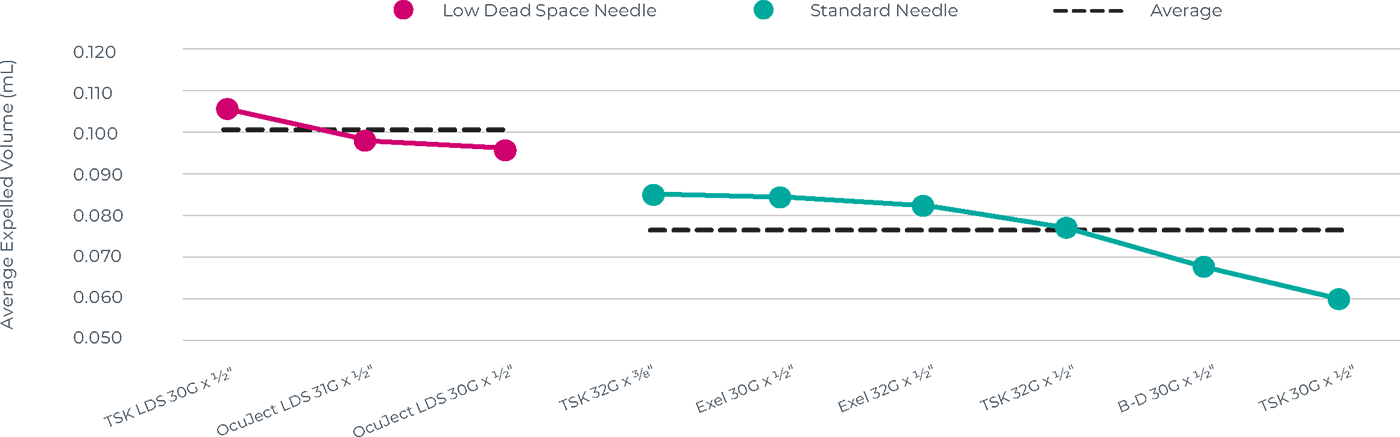
Leiters Health recognizes that a physician’s choice of syringe and injection needles has an impact on dosing accuracy due to the retained fluid in the needle/syringe combination. Due to variable fluid retention (i.e. dead space), the volume of Avastin that is delivered to the patient depends on the combination of syringe, fill volume, and needle selection.
To better inform our customers, Leiters Health polled retina customers to determine the most used needles for intravitreal injections and conducted extensive internal testing of these needles with Leiters Health’s repackaged Avastin and the StaClear syringe.
View Internal Test Results
Needle Data Table
| FILL QUANTITY = 0.11 mL | EXPRESSED NEEDLE VOLUME OF AVASTIN (mL) | ||||
|---|---|---|---|---|---|
| NEEDLE NAME/MANUFACTURER | NEEDLE SIZE | NEEDLE TYPE | MINIMUM | MAXIMUM | MEAN |
| B-D PrecisionGlide™ | 30G x 1/2″ | Standard | 0.059 | 0.078 | 0.069 |
| Exelint International Exel® | 30G x 1/2″ | Standard | 0.071 | 0.092 | 0.086 |
| Exelint International Exel® | 32G x 1/2″ | Standard | 0.077 | 0.102 | 0.082 |
| TSK STERiJECT | 30G x 1/2″ | Standard | 0.058 | 0.070 | 0.062 |
| TSK STERiJECT | 32G x 1/2″ | Standard | 0.059 | 0.089 | 0.078 |
| TSK STERiJECT | 32G x 3/8″ | Standard | 0.072 | 0.094 | 0.086 |
| OcuJect OCUSAFE® | 30G x 1/2″ | Low dead space | 0.091 | 0.104 | 0.098 |
| OcuJect OCUSAFE® | 31G x 1/2″ | Low dead space | 0.074 | 0.106 | 0.100 |
| TSK LOW DEAD SPACE | 30G x 1/2″ | Low dead space | 0.086 | 0.112 | 0.105 |
Data Needle Chart
Leiters Health recently sat down with Dr. Tanya Ghosh, an ophthalmologist from the Munnerlynn Eye Institute. Dr. Ghosh shared her passion for ophthalmology and the importance of high-quality repackaged Avastin for her patients and her retina practice.
Additional Resources
Industry Bulletin
In January of 2018, the FDA issued its final guidance on the repackaging of biological products. This guidance required FDA-registered outsourcing facilities to develop new methods for repackaging Avastin, a vital medication for the treatment of retinal diseases. This Industry Bulletin describes the background behind the FDA’s action and chronicles one 503B outsourcing facility’s journey to meet the guidance.
Certificate of Analysis (COA)
Leiters Health provides a Certificate of Analysis (COA) for all of our products. The COA certifies the identity, strength, quality and purity of a specific lot of compounded or repackaged pharmaceutical product. The COA provides documented assurance that the pharmaceutical product meets the required quality standards and specifications as it relates to sterility, post sterilization endotoxin, clarity, color, visible particulates, subvisible particulates, potency, ID, impurities, pH, and osmolality, as applicable. Hard copies of COAs are provided with every product shipment and COAs can also be accessed electronically through the Leiters Online Ordering Portal.
StaClear Website
To learn more about the StaClear syringe, please visit their website.
Ready to place an order?
Reliability with Reserved Inventory
Leiters Health sources Avastin so you don’t have to. We streamline and simplify your supply chain by securing the inventory you need to flex with your practice demand, and can allow your staff to focus on patient care rather than managing two supply chains.
Benefits include:
- We produce and maintain a 30-day inventory of repackaged Avastin syringes so you can flex up and down your supply with your patient schedule.
- Same day shipping for orders received by 2pm MST.
Increased Access and Convenience with Leiters Online Ordering Portal
The Leiters Online Ordering Portal (LOOP) is an interactive, online tool providing a convenient way to order and manage the medications you purchase from us.
Benefits include:
- Convenient online ordering 24 hours a day / 7 days a week.
- Access detailed account and product information.
- Establish automated subscriptions and ordering templates.
- Learn more about LOOP
Footnotes/References
Avastin® is a registered trademark of Genentech, Inc.
StaClear® is a registered trademark owned by TriboFilm Research, Inc.
1 Mixing, Diluting, or Repackaging Biological Products Outside the Scope of an Approved Biologics License Application Guidance for Industry; https://www.fda.gov/downloads/drugs/guidances/ucm434176.pdf
2 https://www.staclear.com
3 BUD is from the date the drug is compounded or repackaged.
